Engineering a fully autologous iPSC-derived airway-on-a-chip (iAirway-Chip) to model patient-specific pulmonary barrier function and inflammatory responses.

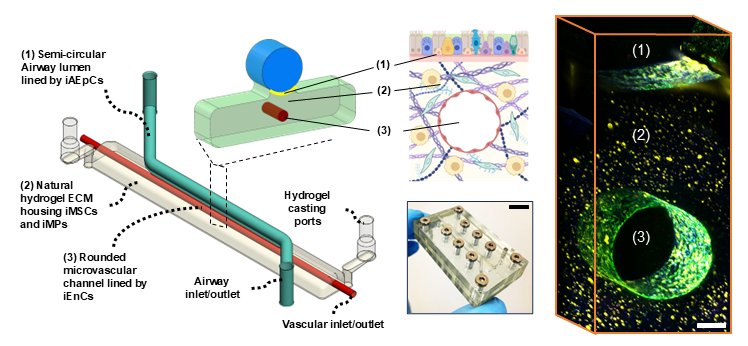
We are developing a fully autologous, immune-competent human airway-on-a-chip platform derived entirely from patient-specific induced pluripotent stem cells (iPSCs) to model pulmonary disease mechanisms in Down syndrome (trisomy 21). The platform reconstructs key structural and cellular features of the human conducting airway by integrating mucociliated airway epithelium, perfusable microvascular endothelium, mesenchymal stromal cells, and mononuclear phagocytes within a thermoplastic microfluidic device containing an extracellular matrix–embedded vascular niche. Using matched disomic and trisomy-21 donor cells, the system reproduces essential airway functions including ciliary beating, epithelial and endothelial barrier integrity, and multicellular immune interactions. Upon influenza infection, the trisomy-21 airway chips exhibit higher viral replication and exaggerated inflammatory signaling, with strong induction of cytokine and interferon-related pathways that mirror clinical observations in individuals with Down syndrome. These results demonstrate the potential of the Next-Generation iAirway-Chip as a personalized microphysiological system for studying genetic disease mechanisms and for accelerating therapeutic discovery in pulmonary disorders.